The Emergent Microbiome: A Revolution for the Life Sciences – Part XIII, The Microbiome and Antibiotic Resistance
Oct 3rd, 2017 by David Puleo | News | Recent News & Articles | The Emergent Microbiome Series |
Antibiotic resistance is a major problem in the United States and is considered by the World Health Organization (WHO) to be one of the largest threats to human health. The top 12 bacterial threats classified by WHO are shown in Table 21. Researchers are going so far as to send antibiotic resistant bacteria into space in order to determine how the bacteria mutate, with the thought being that in space, bacteria will mutate at an accelerated rate, making it easier to study bacterial resistance patterns and, therefore, develop better antibiotics that are active against resistant bacterial forms.
Government-run institutions are investing significantly in the study of antibiotic resistance and resistance mechanisms. For example, in early 2015, the White House released its “National Action Plan For Combating Antibiotic-Resistant Bacteria”. Similarly, in 2016, the CDC passed the Antibiotic Resistance Solutions Initiative, allocating roughly $160 Million in order to “detect, respond, and contain resistant infections across healthcare setting and communities.”2 The CDC also awarded $14 Million to 34 different projects that will further investigate antibiotic resistance as it relates to the microbiome. Yale University is one of the institutions to which funds were given. This article will review antibiotic resistance, how antibiotics affect the microbiome, and those therapeutic strategies being used to prevent or correct microbiome imbalances. Furthermore, we will review some of the significant players and patents in this area.
Problems with Antibiotic Use
Research published in the Journal of the American Medical Association (JAMA 2016, 315, 1864-73) suggests that antibiotics were prescribed unnecessarily in roughly 30% of patient cases. This unwarranted exposure to antibiotics, as well as the misuse, overuse, or incorrect prescription of antibiotics, increases the likelihood of developing resistance. Specifically, the majority of nosocomial (hospital-related) infections are caused by so-called ESKAPE pathogens (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species). These “opportunistic infections” are especially deleterious for patients who have weakened immune systems, e.g. cancer patients undergoing chemotherapy.
The emergence of resistance can occur via different mechanisms which include, but are not limited to (1) production of efflux, or multi-drug resistance (mdr), pumps that, as the name implies, “pump” out antibiotics that are harmful to the organism; (2) production of enzymes that modify antibiotics and, thus, inactivate them or make them less efficacious; and/or (3) mutation of those enzymes that are the targets of antibiotics, thus allowing those enzymes to evade antibiotics.3 The end result is that those resistant bacteria are able to colonize the host, flourish, and overtake host commensal microbiota.
Combatting Clostridium difficile infection (CDI)
Typically, broad-spectrum antibiotics are prescribed for un-diagnosed conditions as a prophylactic, or preventative, measure. However, it is common that treatment with broad-spectrums can wipe out an individual’s healthy gut microflora and cause microbiome imbalance, or dysbiosis. This may further predispose patients to infections from MRSA (Methicillin-resistant Staphylococcus aureus), CRE (carbapenem-resistant Enterobacteriaceae), or Clostridium difficile. Clostridium difficile infection (CDI) is a relatively common and refractory infection, often acquired in hospitals and nursing homes by susceptible elderly patients. The standard-of-care treatment for CDI is usually treatment with other antibiotics, firstly metronidazole, and if that is not efficacious, vancomycin. However, one concern with treatment using vancomycin is the emergence of vancomycin-resistant bacteria.
Rather than treating patients with antibiotics, clinicians are turning to a procedure known as fecal microbiota transplant (FMT), whereby fecal bacteria are transferred from a healthy individual to a recipient patient. The transplant is meant to replenish bacteria that have been depleted from antibiotic treatment in the recipient and, thus, restore eubiosis. An increased number of patents in this space reflect this trend. For example, US9511099 refers to the use of a similar methodology, termed Microbiota Restoration Therapy (MRT). Although mainly used for the treatment of CDI, the patent application also discloses colon cancer. The company that filed the patent application, Rebiotix, has several therapies in clinical trials for CDI treatment and prevention. Referenced briefly in Part V of the Microbiome Series, patents US9028841 and US9446080 from Seres Therapeutics were included below, which also disclose methods for the treatment and prevention of recurrent CDI. Seres’s current lead, SER-109, is currently in Phase II clinical trials and has been reviewed earlier here by Dilworth IP.
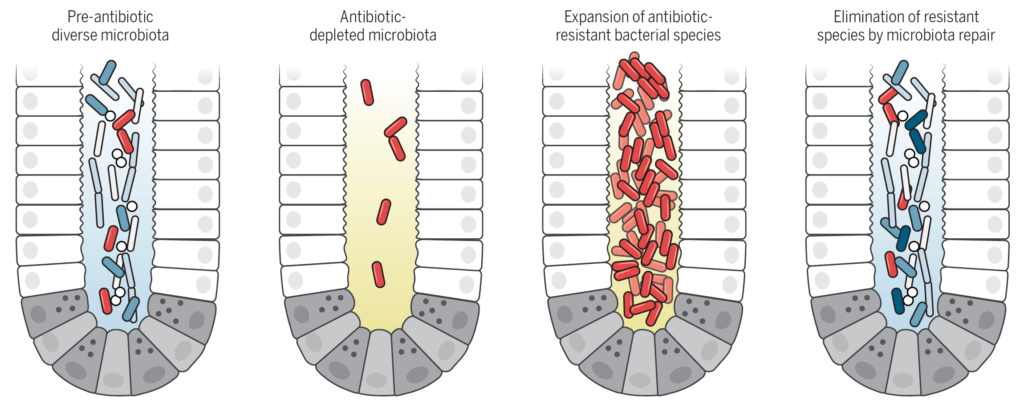
Antibiotic treatment eliminates many commensal bacterial species from the gut lumen and reduces antimicrobial defenses. Antibiotic-resistant bacterial species can thrive, undergo expansion, and traverse the intestinal epithelial barrier. Antibiotic-induced microbiota defects and the resulting loss of colonization resistance can be corrected by administration of commensal bacterial species [Figure and Legend from Science 352, 535-538 (2016)].
Another company, OpenBiome (The Microbiome Health Research Institute), is a non-profit organization that is the first public stool bank. It has patented a capsule containing microbes for the treatment of gastrointestinal disorders (WO2016178775). OpenBiome is also collaborating with Finch Therapeutics on a treatment for CDI.
Alternatives to Antibiotics: Using Immunotherapies to Alter the Microbiome
Although some companies are still focusing their efforts on identifying new antibiotics, others have begun undertaking new approaches. As mentioned above, in the case of CDI, missing bacteria can be replenished via FMT and, thus, restore a patient to eubiosis. However, is it possible to remove a specific bacterium from the host commensal population, which would completely eliminate the need for antibiotics? Numerous companies are attempting just that. Using their proprietary Cloudbreak™ Immunotherapy Platform, Cidara Therapeutics can selectively target and clear gram-negative bacteria. Although no patent directed to the Cloudbreak™ Platform could be found, Cidara has patent application US20160213742 for the treatment of fungal infections, specifically from Aspergillus fumigatus and Candida albicans.
MedImmune, the biologics arm of Astra Zeneca, has patented several monoclonal antibodies (mAbs) targeting bacterial-specific antigens as a means of selectively clearing bacteria. Patent US9527905 claims the use of a mAb targeting Staphylococcus aureus α toxin, a component that enhances the proliferation of this Gram-negative bacteria. The in vivo efficacy of one such mAb, MEDI4893, was described in a 2016 article in Science Translational Medicine [Sci Transl Med 2016, 8, 329ra31]. Another patent application from Arsanis Biosciences, US20150086539, and patent from XBiotech, US9486523, detail mAbs targeting S. aureus. Patent application US20150284450 describes a bispecific mAb targeting Pseudomonas aeruginosa. The clinical candidate, MEDI3902, is detailed in the following: Sci Transl Med 2014, 6, 262ra155. Another anti-P. aeruginosa mAb is detailed in patent US9403901. A mAb that binds the outer membrane of Acinetobacter baumannii is detailed in patent US8747846. An anti-Streptococcus pneumoniae mAb is detailed in patent US9279815.
Moving Forward to Combat Resistance
While seemingly attractive years ago, the use of broad-spectrum antibiotics is now being questioned. A recent paper in Nature Communications (Nat. Commun. 2017, 8, 15062) posits that the use of antibiotics in infants significantly changes their microbiome composition later in life. Thus, in the short term, determining if and when a patient needs antibiotics will help to limit the spread of resistance. Although the aforementioned mAb therapeutics may in fact be superior to standard-of-care broad-spectrum antibiotics, whether and how resistance will develop is still a question. Similarly, the insurance dynamics and cost to the patient may be a significant concern. As new therapies develop, patent protection will certainly be an integral part of the process.
Table 1: Patents and Patent applications
| Patent Number | Inventor | Assignee | Title | Issue or Publication Date |
| US9511099 | Jones, et al. | Rebiotix, Inc. | Microbiota restoration therapy (MRT), compositions and methods of manufacture | Dec 6, 2016 |
| US9028841 | Henn, et al. | Seres Health, Inc. | Synergistic bacterial compositions and methods of production and use thereof | May 12, 2015 |
| US9446080 | McKenzie, et al. | Seres Therapeutics, Inc. | Compositions and methods | Sep 20, 2016 |
| WO2016178775 | Smith, et al. | Microbiome Health Research Institute Inc. | Compositions and methods for therapeutic delivery of microbial communities | Nov 10, 2016 |
| US9403901 | Digiandomenico, et al. | Medimmune, Llc, Medlmmune Limited | Anti-pseudomonas Psl binding molecules and uses thereof | Aug 2, 2016 |
| US20150086539 | Nagy, et al. | Arsanis Biosciences Gmbh | Cross-reactive staphylococcus aureus antibody | Mar 26, 2015 |
| US20160213742 | Forrest, et al. | Cidara Therapeutics, Inc. | Compositions and methods for the treatment of fungal infections | Jul 28, 2016 |
| US20150284450 | Digiandomenico, et al. | Medimmune, Llc, Medlmmune Limited | Combination therapies using anti-pseudomonas psl and pcrv binding molecules | Oct 8, 2015 |
| US8747846 | Spellberg, et al. | Los Angeles Biomedical Research Institute At Harbor-UCLA Medical Center | Compositions and methods for immunization against drug resistant Acinetobacter baumannii | Jun 10, 2014 |
| US9279815 | Kenneth Smith | Oklahoma Medical Research Foundation | Human Streptococcus pneumoniae antibodies and uses therefor | Mar 8, 2016 |
| US9527905 | Sellman, et al. | Medimmune, Llc, | Antibodies that specifically bind staphylococcus aureus alpha toxin and methods of use | Dec 27, 2016 |
| US9486523 | John Simard | Xbiotech, Inc. | Compositions and methods for treating and preventing Staphylococcus aureus infections | Nov 8, 2016 |
Table 2: World Health Organization (WHO) Classification of Top 12 Bacterial Threats1
| WHO Classification | Bacterium | Gram Strain | Antibiotic Resistance | Other Properties |
|
Priority 1: Critical |
Acinetobacter baumannii | Negative | Carbapenem | |
| Pseudomonas aeruginosa | Negative | Carbapenem | ||
| Enterobacteriaceae | Negative | Carbapenem | Produces extended-spectrum-β-lactamases (ESBL) | |
|
Priority 2: High |
Enterococcus faecium | Positive | Vancomycin | |
| Staphylococcus aureus | Positive | Methicillin; Vancomycin | ||
| Helicobacter pylori | Negative | Clarithromycin | ||
| Campylobacter spp. | Negative | Fluoroquinolone | ||
| Salmonellae | Negative | Fluoroquinolone | ||
| Neisseria gonorrhoeae | Negative | Cephalosporin; Fluoroquinolone | ||
|
Priority 3: Medium |
Streptococcus pneumoniae | Positive | Penicillin-non-susceptible | |
| Haemophilus influenzae | Negative | Ampicillin | ||
| Shigella spp. | Negative | Fluoroquinolone |
1 http://www.who.int/mediacentre/news/releases/2017/bacteria-antibiotics-needed/en/
2 https://www.cdc.gov/drugresistance/solutions-initiative/
3 https://www.cdc.gov/getsmart/community/about/antibiotic-resistance-faqs.html
-David Puleo and Anthony D. Sabatelli, PhD, JD
This article is for informational purposes, is not intended to constitute legal advice, and may be considered advertising under applicable state laws. The opinions expressed in this article are those of the author only and are not necessarily shared by Dilworth IP, its other attorneys, agents, or staff, or its clients.