Nanomedicine: A Vast Horizon on a Molecular Landscape – Part IX, Organs-on-a-chip II
Apr 11th, 2017 by Jing Zhou | News | Recent News & Articles |
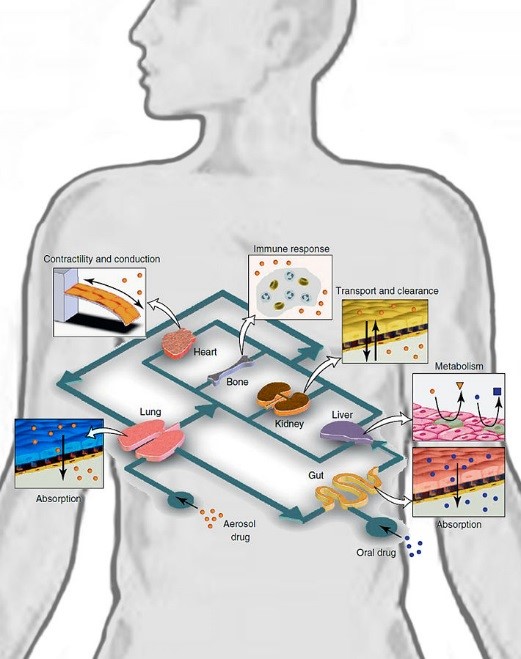 This is the ninth article in a review series on Nanomedicine. We reviewed the major research and entrepreneurial development of nanomedicine and the relevant patent landscape (Part I and Part II). The first topic we discussed was Organs-on-a-chip (Part III). Here, we continue our discussion in this field with focus on entrepreneurial developments. We also have other reviews about nanoparticles for drug delivery (Part IV), cancer therapeutics (Part V), and bio-imaging (Part VI). We also included a discussion about functional nanoparticles: quantum dots (Part VII) and magnetic nanoparticles (Part VIII). As in the past, those patent documents cited in the article are summarized in a table at the end.
This is the ninth article in a review series on Nanomedicine. We reviewed the major research and entrepreneurial development of nanomedicine and the relevant patent landscape (Part I and Part II). The first topic we discussed was Organs-on-a-chip (Part III). Here, we continue our discussion in this field with focus on entrepreneurial developments. We also have other reviews about nanoparticles for drug delivery (Part IV), cancer therapeutics (Part V), and bio-imaging (Part VI). We also included a discussion about functional nanoparticles: quantum dots (Part VII) and magnetic nanoparticles (Part VIII). As in the past, those patent documents cited in the article are summarized in a table at the end.
Organs-on-a-chip
Recently, Draper announced a three-year agreement with Pfizer. This collaboration focuses on developing effective disease models for testing potential drug candidates based on microphysiological systems, also known as “organs-on-a-chip”.
The organs-on-a-chip technology is a three-dimensional microfluidic based multi-cell co-culture system that models the physiological, mechanical, and molecular environment of the human body and mimics the physiological functions of human organs. This technology offers unique in vitro disease models for new drug screening and toxicology testing. This technology has attracted attentions not only from academic institutes but also from the pharmaceutical industry. One of the main reasons for this interest is the potential cost and time savings for drug research and the development process. As required by the FDA drug approval process, new drug chemical entities are tested in animals before going into human Phase I testing for the drug approval process. The preclinical animal testing process is tedious and extremely expensive. Additionally, animal models are not always predictive for characterizing drug safety in humans. About 40% of drug compounds fail in Phase I clinical trials (Clinical Development Success Rates 2006-2015, BIO Industry Analysis, June 2016). To address these challenges, organs-on-a-chip has been proposed as a novel method to develop human disease models and replace preclinical animal testing.
We have briefly reviewed the research development and IP landscape in organs-on-a-chip. Here we would like to focus on the entrepreneurial developments in this field. As in the past, those patent documents cited in the article are summarized in the table at the end.
AxoSim Technologies
AxoSim is a New Orleans based startup launched in 2014. Its main pipeline is a Nerve-On-A-Chip, which is a 3D cell-based model mimicking living nerve tissue. It aims at preclinical prediction of neurological safety and efficacy in the early stages of drug development. This technology was developed from Dr. Michael J. Moore’s group at Tulane University (US 20150112244).
Draper
The Charles Stark Draper Laboratory is an American not-for-profit research and development organization, having a long history from 1932. In 2009, Draper initiated a new area of medical systems. Draper closely collaborated with scientists at MIT to develop microphysiological systems to emulate human organs and create disease models. At the end of 2016, Draper announced a 3-year agreement with Pfizer, using the “organs-on-a-chip” technology to facilitate pre-clinical drug development with a focus on personalized medicine. Currently Draper has built three microphysiological systems for modeling liver, vasculature and gastrointestinal organs (US 7,670,797, US 8,951,302, US 9,067,179, US 9,528,082).
Emulate Bio
Emulate Bio is a Wyss Institute spin-off company launched in 2014. It focuses on developing multiple organ-on-a-chip systems to model human physiological systems. The technology is based on discoveries in Dr. Donald Ingber’s lab, using models of the lung, liver, intestine, skin and brain (US 8,647,861). This lab is also interested in other organ systems such as the kidney and heart. In 2015, Emulate collaborated with Johnson & Johnson and Merck using organs-on-a-chip for drug discovery and development processes. In 2016, Emulate announced a collaboration with Seres Therapeutics to investigate Novel Microbiome Therapeutics for inflammatory Bowel Disease.
Hepregen
Hepregen is a MIT spin-off company founded in 2007, based on a technique developed in Dr. Sangeeta Bhatia’s lab (US 6,133,030). Its main product, HepetoPac Assay, utilizes a micropatterned hepatocyte co-culture system to model the metabolic activities of a liver system and was released in 2013. Their other pipeline product is HepetoMune, targeting an inflamed human liver model.
HμREL
HμREL is a Merck supported company, which was incubated in New Jersey from 2007-2011. Its technology originated from Dr. Michael Shuler’s group at Cornell University (US 7,288,405 and US 8,748,180). In 2013, HμREL launched three liver-on-a-chip products for human, rat and dog. HμREL has also established a collaboration with Sanofi for pre-clinic drug development.
InSphero
InSphero is a Swiss company founded in 2009. They use a scaffold-free 3D cell culture technique to generate self-assembled microtissues, emulating human organ systems (US 9,267,103 and WO/2017/001680). Their current pipelines include liver, pancreas, tumor, and skin microtissue systems and in vitro toxicology and drug discovery services.
Nortis
Nortis is a Seattle based company, spun out of the University of Washington in 2012. Nortis developed a microfluidic kidney-on-a-chip for drug testing and launched its commercial product on 2015 (US 7,622,298 and US 20150240194A1).
Tara Biosystems
Tara Biosystems is a New York-based Columbia University spin out company founded in 2014. Their focus is on developing a “heart-on-a-chip” system. The technology is based on research from Dr. Gordana Vunjak-Novakovic’s group at Columbia University and Dr. Milica Radisic’s group at Toronto University (US 20170002330A1 and US 20160282338). Tara Biosystems uses a Biowire platform, to introduce electrical stimulation on a microchip to stimulate stem cells to mature into heart tissue. This microtissue mimics adult heart muscles, offering a platform for drug discovery, cardiac toxicology, and personalized cardiology.
TissUse
TissUse is a Berlin, Germany-based company developing a “Multi-Organ-Chip” platform based on technology discovered in Dr. Roland Lauster’s lab at Technische Universitat Berlin (US 20130295598). This company uses a multi-organ-chip as a platform to emulate human metabolic activities and accelerate the development of pharmaceutical, chemical, cosmetic, and personalized medical products. Currently, TissUse has announced their 2-Organ-Chip and 4-Organ-Chip products, involving simultaneously culturing from 2 to 4 different organ equivalents on a single chip connected to each other by perfusion channels or vasculature. Their next goal is to develop a human-on-a-chip system, with a larger number of organs cocultured on a single chip.
| Patent Number | Title | Assignee | Inventors |
| US 20150112244 | SELECTIVELY POLYMERIZABLE COMPOSITIONS AND METHODS OF USE IN VIVO | The Administrators of the Tulane Educational Fund | Elaine Horn-Ranney; Parastoo Khoshakhlagh; Michael Moore; Jesse Ranney |
| US 7,670,797 | Method of determining toxicity with three dimensional structures | The General Hospital Corporation;
The Charles Stark Draper Laboratory |
Joseph P. Vacanti; Robert Rubin; Wing Cheung; Jeffrey T.Borenstein
|
| US 8,951,302 | Biomimetic vascular network and devices using the same | The General Hospital Corporation;
The Charles Stark Draper Laboratory |
Howard I. Pryor; Ira Spool; David M. Hoganson; Joseph P. Vacanti; Jeffrey T.Borenstein |
| US 9,067,179 | Microfluidic device facilitating gas exchange, and methods of use and manufacture thereof | The Charles Stark Draper Laboratory, Inc. | Joseph L. Charest; Jeffrey T. Borenstein; Joseph M. Bauer
|
| US 9,528,082 | Modular platform for multi-tissue integrated cell culture | The Charles Stark Draper Laboratory, Inc. ;
Massachusetts Institute of Technology |
Joseph Cuiffi; Jeffrey T. Borenstein; Anilkumar Harapanahalli Achyuta; Mark J. Mescher; Linda Griffith; Samuel Walker Inman |
| US 8,647,861 | Organ mimic device with microchannels and methods of use and manufacturing thereof | Children’s Medical Center Corporation | Donald E. Ingber; Dongeun Huh
|
| US 6,133,030 | Co-cultivation of cells in a micropatterned configuration | The General Hospital Corporation;
Massachusetts Institute of Technology |
Sangeeta Bhatia; Martin Yarmush; Mehmet Toner |
| US 7,288,405 | Devices and methods for pharmacokinetic-based cell culture system | Cornell Research Foundation, Inc. | Michael Shuler; Gregory T. Baxter; Aaron Sin; Robert Andrew Harrison; Scott Meyers |
| US 8,748,180 | Microfluidic device for pharmacokinetic-pharmacodynamic study of drugs and uses thereof | Cornell University | Michael L. Shuler; Jong Hwan Sung
|
| US 9,267,103 | Compliant multi-well plate | INSPHERO AG | Jan Lichtenberg; Wolfgang Moritz; Jens Kelm |
| WO/2017/001680 | DEVICE FOR PROPAGATING MICROTISSUES | INSPHERO AG | Jan LICHTENBERG;
Wolfgang MORITZ; Jens KELM |
| US 7,622,298 | Method for creating perfusable microvessel systems | Norits, Inc.
|
Thomas Neumann |
| US 20150240194A1 | Microfluidic System for Reproducing Functional Units of Tissues and Organs In Vitro | NORTIS, INC. | Thomas Neumann; Anna A. Tourovskaia; Mark E. Fauver; Greg Kramer; Elizabeth Asp; Henning Mann |
| US 20170002330A1 | ENGINEERED ADULT-LIKE HUMAN HEART TISSUE | THE TRUSTEES OF COLUMBIA UNIVERSITY IN THE CITY OF NEW YORK | Gordana Vunjak-Novakovic; Keith Yeager; Kacey Ronaldson; Stephen Ma; Timothy Chen |
| US 20160282338 | COMPOSITIONS AND METHODS FOR MAKING AND USING THREE-DIMENSIONAL ISSUE SYSTEMS | Jason Miklas; Milica Radisic; Nimalan Thavandiran; Sara Vasconcelos; Yun Xiao; Boyang Zhang; Yimu Zhao | |
| US 20130295598 | CIRCULATION SYSTEM | TECHNISCHE UNIVERSITAET BERLIN | Uwe Marx; Gerd Lindner; Reyk Horland; Silke Hoffman; Roland Lauster;
|
– Jing Zhou, PhD and Anthony D. Sabatelli, PhD, JD
This article is for informational purposes, is not intended to constitute legal advice, and may be considered advertising under applicable state laws. The opinions expressed in this article are those of the author only and are not necessarily shared by Dilworth IP, its other attorneys, agents, or staff, or its clients.
